 Terbium has a simple ferromagnetic ordering at temperatures below 219 K. Single terbium atoms have been isolated by implanting them into fullerene molecules. Terbium easily oxidizes, and is therefore used in its elemental form specifically for research. The yttrofluorite variety of the mineral fluorite owes its creamy-yellow fluorescence in part to terbium. The terbium(III) cation is brilliantly fluorescent, in a bright lemon-yellow color that is the result of a strong green emission line in combination with other lines in the orange and red. Only three electrons can be removed before the nuclear charge becomes too great to allow further ionization, but in the case of terbium, the stability of the half-filled 4f 7 configuration allows further ionization of a fourth electron in the presence of very strong oxidizing agents such as fluorine gas. The eleven 4f and 6s electrons are valence. The 65 electrons of a terbium atom are arranged in the electron configuration 4f 96s 2. Terbium exists in two crystal allotropes with a transformation temperature of 1289 ☌ between them. It is relatively stable in air compared to the earlier, more reactive lanthanides in the first half of the lanthanide series. Terbium is a silvery-white rare earth metal that is malleable, ductile and soft enough to be cut with a knife. Terbium green phosphors are combined with divalent europium blue phosphors and trivalent europium red phosphors to provide trichromatic lighting technology, a high-efficiency white light used for standard illumination in indoor lighting.Ĭharacteristics Physical properties Terbium oxide is used in fluorescent lamps and television and monitor cathode-ray tubes (CRTs). Most of the world's terbium supply is used in green phosphors. As a component of Terfenol-D (an alloy that expands and contracts when exposed to magnetic fields more than any other alloy), terbium is of use in actuators, in naval sonar systems and in sensors. Terbium is used to dope calcium fluoride, calcium tungstate and strontium molybdate in solid-state devices, and as a crystal stabilizer of fuel cells that operate at elevated temperatures. 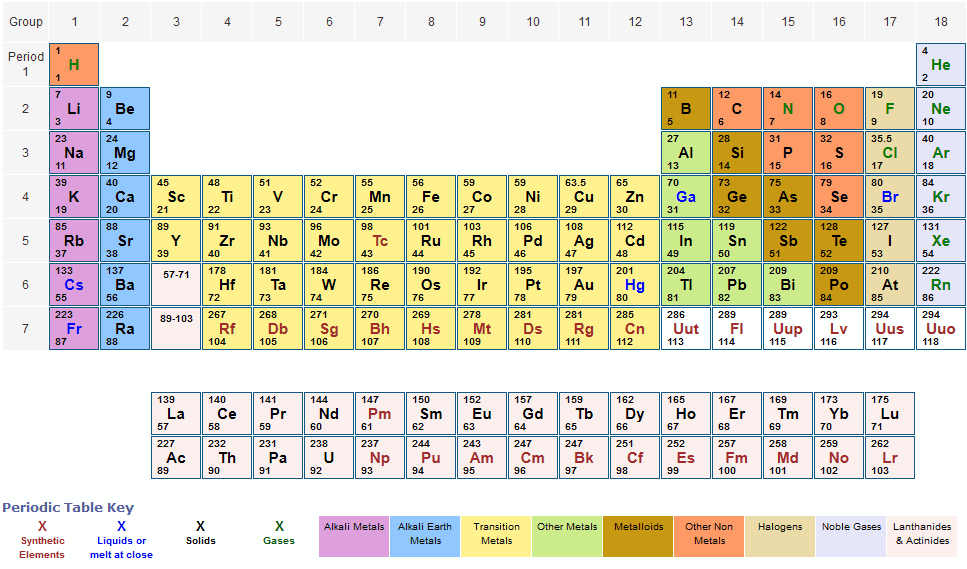
Terbium was not isolated in pure form until the advent of ion exchange techniques. Yttrium and terbium, as well as erbium and ytterbium, are named after the village of Ytterby in Sweden. He detected it as an impurity in yttrium oxide, Y 2O 3. Swedish chemist Carl Gustaf Mosander discovered terbium as a chemical element in 1843. Terbium is never found in nature as a free element, but it is contained in many minerals, including cerite, gadolinite, monazite, xenotime and euxenite. The ninth member of the lanthanide series, terbium is a fairly electropositive metal that reacts with water, evolving hydrogen gas. It is a silvery-white, rare earth metal that is malleable, and ductile. 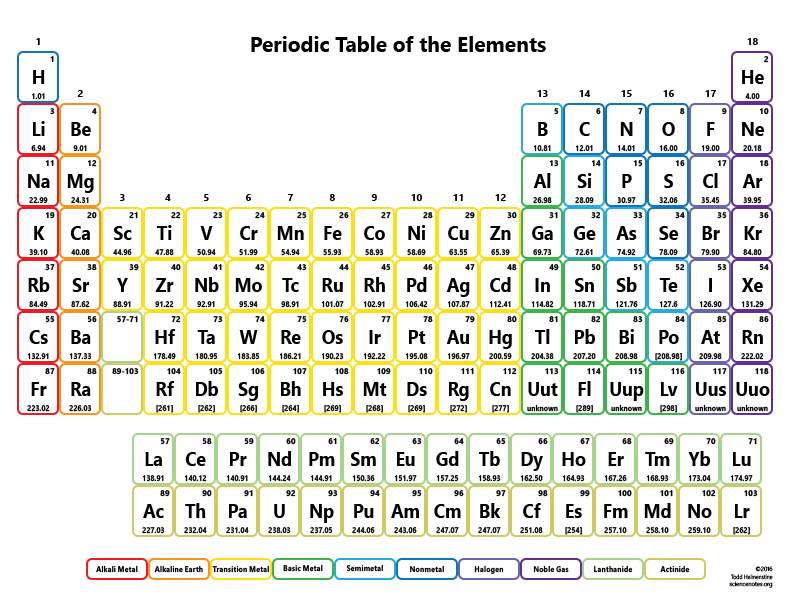
Terbium is a chemical element it has symbol Tb and atomic number 65. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
